
 There are many sources of misunderstanding in matters related to science and belief in God. In this journal we try to explore some of the misunderstandings that we feel qualified to discuss. There are many areas that I do not feel qualified to deal with either because I lack education in those areas or because I am still struggling with my own understandings. In all areas of science this is the case, and it is not just the limits of an individual that is involved. Man’s understanding of very basic things has evolved through the years to where it is today, and this evolution continues.
There are many sources of misunderstanding in matters related to science and belief in God. In this journal we try to explore some of the misunderstandings that we feel qualified to discuss. There are many areas that I do not feel qualified to deal with either because I lack education in those areas or because I am still struggling with my own understandings. In all areas of science this is the case, and it is not just the limits of an individual that is involved. Man’s understanding of very basic things has evolved through the years to where it is today, and this evolution continues.
 A wonderful example of this that has taken place in my lifetime has been what an atom looks like. When I was a child in the 1940s an atom was considered to be a positively charged nucleus made up of protons and neutrons with electrons going around that nucleus in specific orbits.
A wonderful example of this that has taken place in my lifetime has been what an atom looks like. When I was a child in the 1940s an atom was considered to be a positively charged nucleus made up of protons and neutrons with electrons going around that nucleus in specific orbits.
The atom look-ed very much like a solar system and that was the level of understanding I had. I saw diagrams which supported that view. When I learned about the spectra of various elements, my level of understanding increased. The different electron orbits had different energies and the further out from the nucleus they were, the more energy they had. These orbits were given letters — K, L, M, N, etc. Many things in chemistry could be explained by using these orbits. Each level held a specific number of electrons — K had 2, L had 8, M had 18, N had 32, etc.
When I started teaching chemistry in 1959 this level of understanding was what the textbooks presented. As I took graduate work in chemistry and physics, I realized that what I was teaching my students was grossly oversimplified. The questions asked by my better students demonstrated that they saw the problems, too.
During those years research had shown that the atom was mostly empty space. If the nucleus of an atom were the size of a marble, the first location of electrons would be miles away. In addition, the electrons were not like little marbles orbiting a central core. In fact, the electrons had wave properties, and what our drawings actually represented were the probabilities of locating an electron at a certain position. Electrons had magnetic properties and spun on their axes like a top. That meant they strove to be paired and a magnetic field would affect them. Besides that, their paths around the nucleus were not neat circles or ellipses. Some traveled in figure eights, some in clover leafs with spectral names being applied to the shapes:
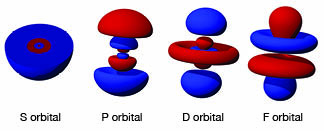
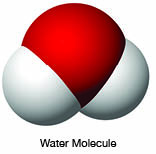 This new level of understanding was wonderful. It explained why water was a polar molecule. Its oxygen atom had a P orbital with electrons arranged at a 90 degree angle between two of the arms so that the two hydrogen atoms that attached to form the water could only be attached at 90 degree angles (bent to 105 degrees by the repulsive force of the two hydrogen atoms). The old K orbit with two electrons was actually a pair of electrons in a spherical orbital. The L orbit was actually two electrons in the second orbit in a spherical orbital, and six electrons in a P orbital.
This new level of understanding was wonderful. It explained why water was a polar molecule. Its oxygen atom had a P orbital with electrons arranged at a 90 degree angle between two of the arms so that the two hydrogen atoms that attached to form the water could only be attached at 90 degree angles (bent to 105 degrees by the repulsive force of the two hydrogen atoms). The old K orbit with two electrons was actually a pair of electrons in a spherical orbital. The L orbit was actually two electrons in the second orbit in a spherical orbital, and six electrons in a P orbital.
The point of this oversimplified discussion is that the level of understanding of the atom has changed as better information has become available. The original understandings were oversimplified, and I would frequently tell my students that.
A similar discussion could be given concerning what science considered light to be. Because light could be broken up into different colors by refraction or diffraction science considered it to be factual that light is a wave. Because waves cannot travel through a vacuum, space  was said to be filled with a substance called “ether” (aether) which allowed light to travel through it. This perception of light is still taught today in many elementary classes. In the late nineteenth and early twentieth century experimental evidence showed that light behaved as a particle. It could knock electrons out of some crystals showing it had momentum, a property of particles. It could pass through a man-made vacuum. It could change energy when it collided with another particle. These are all mass properties. If you were to shine it on a black cloth which absorbed most of the light, the cloth did not gain mass. That did not fit the physics definition of what a particle is. The scientists had to move to a new level of thinking which could accept some things that violated their previous understandings.
was said to be filled with a substance called “ether” (aether) which allowed light to travel through it. This perception of light is still taught today in many elementary classes. In the late nineteenth and early twentieth century experimental evidence showed that light behaved as a particle. It could knock electrons out of some crystals showing it had momentum, a property of particles. It could pass through a man-made vacuum. It could change energy when it collided with another particle. These are all mass properties. If you were to shine it on a black cloth which absorbed most of the light, the cloth did not gain mass. That did not fit the physics definition of what a particle is. The scientists had to move to a new level of thinking which could accept some things that violated their previous understandings.
The two examples from science that we have used have come about as scientists have gained information and understanding. In both of these examples there were reputable scientists who opposed the new understanding for a variety of reasons. Often their objections were merely resistance to something they did not fully understand. The concept seemed to violate their previous understandings and their common experience. Rather than try to achieve a new level of understanding they simply rejected it.
My point is that in both science and faith it is easy for us to reject new evidence which seems to contradict our preconceived notions. Both scientists and Christians are still human and our human prejudices create false conflicts between science and faith. Science and faith are friends — not enemies. (See also “Science Fraud” on page 29.)
Picture credits:
Cover montage and atom illustration (top): Roland Earnst
Illustrations: en.wikipedia.org/wiki/Atomic_orbital
Illustration: en.wikipedia.org/wiki/Water_(molecule)
© visual28. Image from BigStockPhoto.com
